Départements Français
Les divisions ont été fondées le 4 janvier 1790 par l'Assemblée constituante pour annuler les provinces de France considérées comme archaïques, irrationnelles et inégales. Ils doivent éliminer les structures de l'ancien régime et construire une nation homogène. Pour des raisons de rationalité, les départements ont une architecture similaire: une partie du territoire si petite qu’elle peut être facilement gérée par une capitale idéalement située dans le centre. De même, les départements n'étaient pas nommés selon des critères historiques, mais purement géographiques (noms de rivières, de montagnes, etc.). Le nombre de divisions, à l'origine 28, a augmenté avec les réalisations territoriales de la République et de l'Empire et a été réduit à 86 après sa chute en 1815. L'acquisition de Nice et de Savoie en 1860 a porté le total à 89.
Trois départements français d'Alsace et de Lorraine (Haut-Rhin, Bas-Rhin et une partie de la Meurthe et de la Moselle) ont été cédés à l'Allemagne en 1871 et rendus à 1919. Les parties non jointes de la Meurthe et de la Moselle ont été fusionnées en Meurthe et Moselle. . Une partie du Haut-Rhin autour de Belfort, qui est restée française en 1871, n’est plus utilisée dans son département d’origine en 1919 et est devenue le territoire de Belfort en 1922.
La restructuration de la région parisienne (1964) et la division de la Corse (1976) ont ajouté six sections. Avec les quatre départements actuels à l'étranger, le nombre total a été porté à 100.
Le 31 mars 2011, Mayotte devient le cinquième département d'outre-mer.
Les départements sont numérotés par ordre alphabétique à la majorité. L’histoire de la France a toutefois conduit à des ajustements et à des exceptions à la règle.
Le numéro de département est en fait un code à 2 ou 3 chiffres qui est numérique (éventuellement un zéro) ou, exceptionnellement, alphanumérique (par exemple, Corse).
Le code est composé de 2 caractères pour les départements de France métropolitaine et de 3 caractères pour le pays étranger (en plus des départements, les communautés étrangères ont un code).
Le conseil de district est l’assemblée consultative du département en tant que collectif territorial formé par la réunion des conseils départementaux. Par ses délibérations, il réglemente les affaires du ministère dans les domaines de responsabilité que lui attribue la loi. Il se réunit au moins une fois par trimestre. Le conseil de district détermine ses règles de procédure et peut y créer des commissions. La majorité absolue de ses membres est requise pour la validité des délibérations.
2016: Trois nouvelles lois modifient les fonctions des départements: la loi du 28 décembre 2015 sur l'adaptation de la société au vieillissement (entrée en vigueur le 1er janvier 2016), la loi du 26 janvier 2016 sur modernisation de notre système de santé et de santé. Loi du 14 mars 2016 sur la protection de l'enfant.
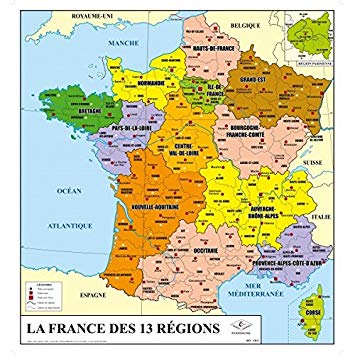
Les régions françaises
L'histoire des régions, associée à la décentralisation, se confond avec la structure de la nation française. Jusqu'à la révolution de 1789, la France comptait déjà 39 districts territoriaux, résultat de conquêtes militaires, d'alliances, d'héritage, de géographie et de peuplements: ils avaient des provinces et des généraux, dont les noms étaient déterminés par les intendants du roi, mais aussi des comtés, des marches, des duchés. Avant que la France atteigne la division actuelle de ses 22 régions, elle coulera pendant près de deux siècles.
Le 16 décembre 2010, à la demande de Nicolas Sarkozy, alors président de la République, le Parlement a approuvé la loi de réforme des collectivités territoriales dans le but de renouveler l'architecture institutionnelle locale afin de réduire le coût du "mile" " territoriale ". Remédier aux faiblesses de l'organisation territoriale et clarifier les responsabilités. Le conseiller territorial, qui aura un seul mandat, sera nommé pour six ans. Ce texte crée une forte résistance entre le Sénat et l’Assemblée nationale et est finalement accepté à une faible majorité. Les dispositions visent également à renforcer et à simplifier l'intercommunauté et à créer une nouvelle catégorie d'institutions publiques de coopération intercommunale: la métropole, qui sera définie en janvier 2014 sous la présidence de François Hollande.